
Despite a number of available medications for the treatment of depression, most patients take many weeks, if not months, to respond to currently approved drugs, and the majority of patients never attain sustained remission of their symptoms. The most exciting development for the pharmacologic treatment of depression in decades is the finding that ketamine has rapid antidepressant effects: a single administration of ketamine to individuals with treatment-resistant depression results in rapid improvement in core depressive symptoms including mood, anhedonia, and suicidal ideation, which is sustained for about a week. However, the widespread clinical use of ketamine is limited due to its abuse potential and capacity to produce dissociative effects.
Ketamine is rapidly and stereospecifically metabolized to norketamine, dehydronorketamine, hydroxyketamine and the hydroxynorketamines (HNKs). We have recently shown (Zanos et al., Nature, 2016) that ketamine’s antidepressant actions in animal models are mediated through a particular hydroxynorketamine metabolite, (2S,6S;2R,6R)-HNK, and in particular the (2R,6R)-HNK stereoisomer, which exerts behavioral, electroencephalographic, electrophysiological and cellular antidepressant-related actions. These antidepressant actions involve sustained activation of AMPA glutamate receptors. We also established that (2R,6R)-HNK lacks ketamine-related side effects in animal models. We are studying the mechanisms underlying these effects, in anticipation that such knowledge will lead to new treatments for depression.
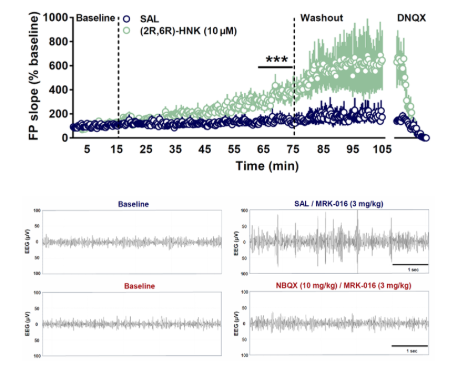
The findings with ketamine indicate that it is possible to develop novel, rapid-acting antidepressants that alleviate symptoms and their underlying pathophysiology within minutes, rather than weeks or months. The laboratory routinely utilizes a number of experimental approaches in order to evaluate the efficacy of therapeutic agents used in the treatment of mood disorders. Some of these include behavioral assays commonly used in preclinical research, as well as electroencephalography (EEG), biochemical, electrophysiological studies in collaboration with other groups. Additionally, we are involved in a number of collaborative translational studies with clinically focused research groups.

The high heritability of bipolar disorder and depression, estimated to be up to 90 and 50 percent respectively, indicates that genetic variation in particular genes predisposes to the development of these illnesses and that identification of the functional relevance of such genetic variation is likely to provide valuable insights that will ultimately lead to new treatments. One such genetic finding in humans is an association between polymorphisms in CACNA1C, a gene that codes for the α1 subunit of the CaV1.2 L-type calcium channel, and a diagnosis of bipolar disorder or depression. We are assessing the impact of CaV1.2 on behavior and other brain functions in the mouse within a context relevant to mood disorder pathophysiology. We are additionally conducting molecular genetic studies using human postmortem brain tissue.Sample text. Click to select the Text Element.
Extensive clinical evidence for the use of lithium as a mood stabilizer, with antimanic, antidepressant, and antisuicidal actions has accumulated over many decades. However, the mechanisms underlying these beneficial effects of lithium are unknown, limiting the potential to identify novel targets that may result in improved therapeutic approaches. We dissect lithium-sensitive behaviors in the mouse as a means to identify the molecular and genetic underpinnings of lithium response. Knowledge derived from these studies may ultimately lead to further optimization of treatment for patients who suffer from mood disorders.
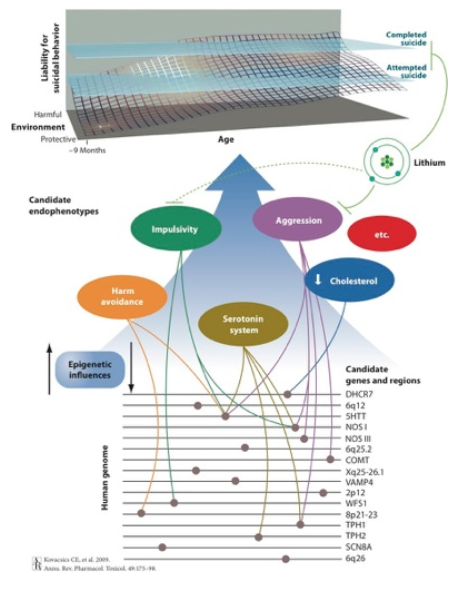
Candidate gene regions, genes, and endophenotypes implicated in a biological systems approach to suicide research. The top portion depicts the dynamic interplay among genetic, environmental, and epigenetic factors that produce cumulative liability to demonstrating thoughts and behaviors related to suicide. Lithium therapy is associated with a decreased risk of suicide attempts and completed suicide, which may be due in part to moderating effects of the drug on aggression and impulsivity.
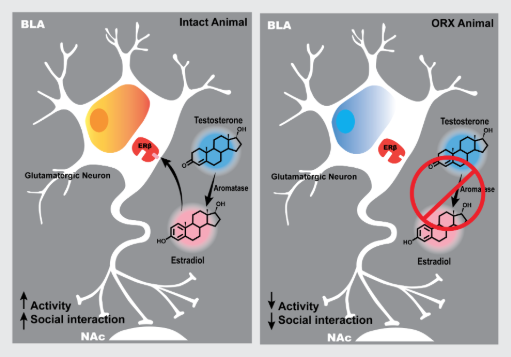
Gonadal hormones, especially estradiol, are suspected to play a role in mood and anxiety disorder pathophysiology. We aim to understand the interactions between underlying genetic susceptibility and gonadal hormones that lead to genetically medicated sex differences. Related projects seek to better understand the therapeutic roles of estradiol in females as well as males.